
Thomson was awarded the 1906 Nobel prize for physics for this work. These particles were later identified to be subatomic particles by J. 
If an object were placed in the tube before the anode end, a shadow would be formed on the glass wall, suggesting some particles travelled from the cathode toward to anode in the tube. However, at the anode end of the tube, the glass wall itself began to glow. Crookes found that this darkspace expanded from the cathode towards the anode as he pumped more air out of the tube until the whole tube was dark. Faraday was the first to notice a dark space just in front of the cathode, where there was no luminescence (known as “cathode dark space”, “Faraday dark space” or “Crookes dark space”). By the 1870s, British physicist William Crookes and others were able to evacuate the tubes to an even lower pressure, below 10 -6 atom. Different elements have different characteristic atomic energy states, thus producing lights of different colors (neon lights). After the strike, electrons within the atoms were excited, and when they returned to lower energy states, energy was released as electromagnetic waves (lights). Of course, we know today that the light arc or glow in these tubes was not electrons, but produced by electrons when they travelled through the tube and struck the residual air atoms. The voltage applied between the two electrodes was anywhere between a few kilovolts and 100 kilovolts and they were called Geissler tubes, similar to today’s neon signs. He observed, instead of an arc, a glow filled the whole tube. In 1857, German physicist and glassblower Heinrich Geissler sucked more air out with an improved vacuum pump, to a pressure of around 10 -3 atom. In 1838, Michael Faraday applied a high voltage through a glass tube filled with rarefied air and noticed a light arc starting from the cathode (the electrode connected to the negative end of the power supply) and ending at the anode (the positive electrode). In 1705, it was already noted that electrostatic generator sparks travel a longer distance through low-pressure air than in atmospheric air. 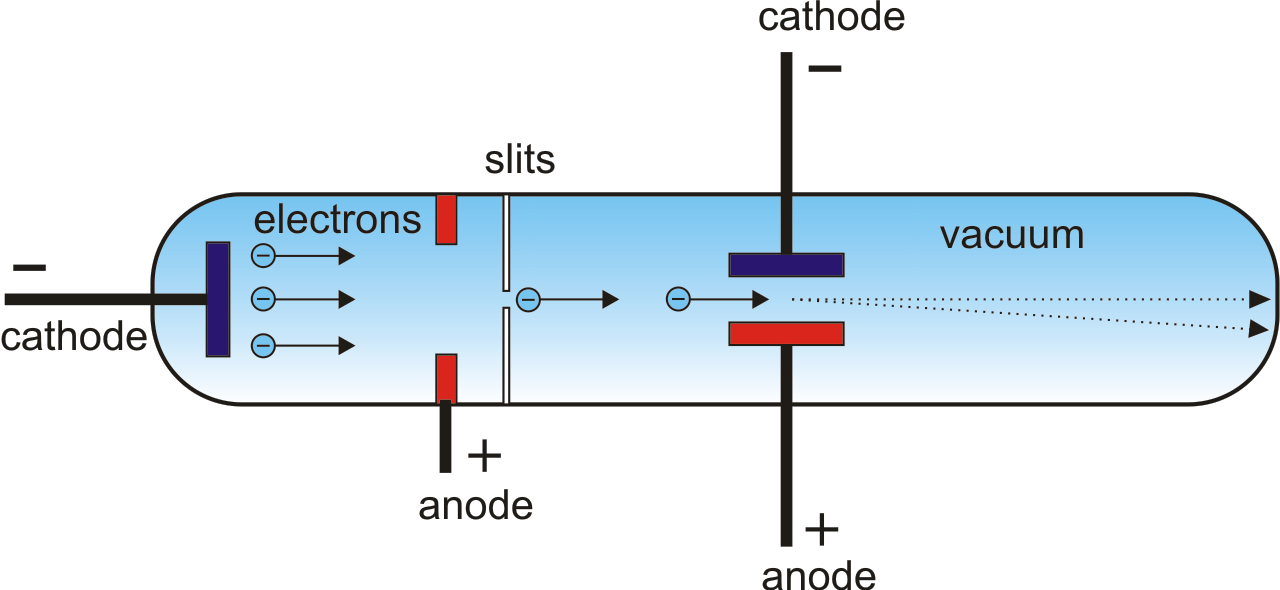
After the 1654 invention of the vacuum pump by Otto von Guericke, physicists began to experiment with passing high voltage through rarefied air. Study of cathode rays began in the early 19 th century, way before the identification of electrons. Such tubes are often referred to as cathode ray tubes. Cathode rays, as we now know, are streams of electrons generated by high electric field-induced gas ionization (old cold cathode ray tubes) or heat-induced thermionic emission (modern vacuum tubes) in specially prepared tubes.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
